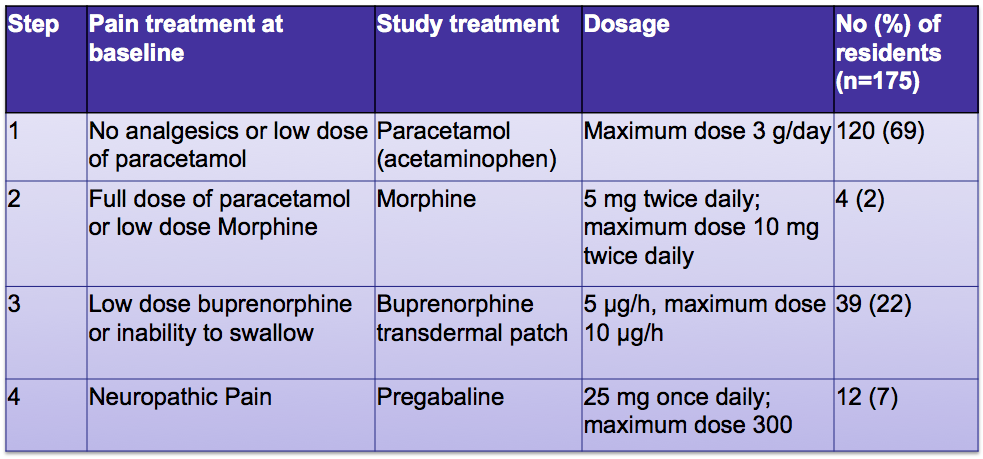
Transdermal rivastigmine for dementia: transdermal formulation may reduce side effects, making optimal dosing easier - Document - Gale Academic OneFile

PDF) Safety and tolerability of transdermal and oral rivastigmine in Alzheimer's disease and Parkinson's disease dementia
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT <invented name> 4.6 mg/24 h transdermal patch <inve