Faster Dissociation: Measured Rates and Computed Effects on Barriers in Aryl Halide Radical Anions | Journal of the American Chemical Society
Kinetics and mechanism of the gasâ•'phase OH hydrogen abstraction reaction from methionine: A quantum mechanical approach

The C4H6•+ Potential Energy Surface. 1. The Ring-Opening Reaction of Cyclobutene Radical Cation and Related Rearrangements | Journal of the American Chemical Society

Cation−π Interaction: Its Role and Relevance in Chemistry, Biology, and Material Science | Chemical Reviews
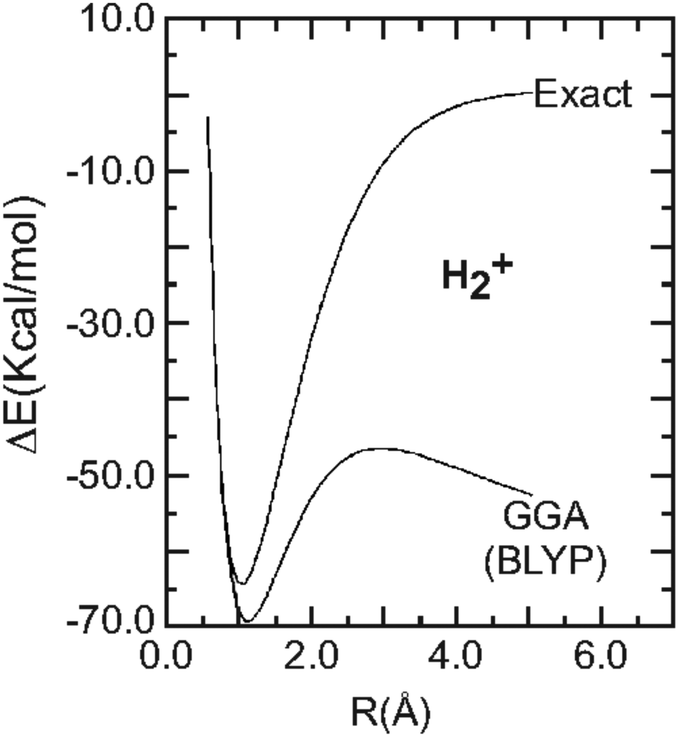
![PDF] Hydrogen Molecule Dissociation Curve with Functionals Based on the Strictly Correlated Regime. | Semantic Scholar PDF] Hydrogen Molecule Dissociation Curve with Functionals Based on the Strictly Correlated Regime. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/44d706e1233349d96782915ff4b7258f2aafc782/14-Figure1-1.png)
PDF] Hydrogen Molecule Dissociation Curve with Functionals Based on the Strictly Correlated Regime. | Semantic Scholar

PDF) Hydrogen bonding between histidine and lignin model compounds or redox mediators as calculated with the DFT method. Effects on the ease of oxidationElectronic supplementary information (ESI) available: Fig. S1: comparison of

PDF) Density functionals that are one- and two- are not always many-electron self-interaction-free, as shown for H-2(+), He-2(+), LiH+, and Ne-2(+)
Piecewise linearity, freedom from self-interaction, and a Coulomb asymptotic potential: three related yet inequivalent properties of the exact density ... - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/D0CP02564J

Stability, Metastability, and Unstability of Three-Electron-Bonded Radical Anions. A Model ab Initio Theoretical Study | Journal of the American Chemical Society

Homolytic versus Heterolytic Dissociation of Alkalimetal Halides: The Effect of Microsolvation - Osuna - 2009 - ChemPhysChem - Wiley Online Library

Mechanism of Formation of Hydrogen Trioxide (HOOOH) in the Ozonation of 1,2-Diphenylhydrazine and 1,2-Dimethylhydrazine: An Experimental and Theoretical Investigation | Journal of the American Chemical Society

PDF) Spin-component-scaled and dispersion-corrected second-order Møller-Plesset perturbation theory: A path toward chemical accuracy

Comparison of the performance of exact-exchange-based density functional methods: The Journal of Chemical Physics: Vol 137, No 11

H2 Dissociation on H-Precovered Ni(100) Surface: Physisorbed State and Coverage Dependence,The Journal of Physical Chemistry C - X-MOL

Direct versus Hydrogen‐Assisted CO Dissociation on the Fe (100) Surface: a DFT Study - Elahifard - 2012 - ChemPhysChem - Wiley Online Library

Homolytic versus Heterolytic Dissociation of Alkalimetal Halides: The Effect of Microsolvation - Osuna - 2009 - ChemPhysChem - Wiley Online Library
How well can density functional theory and pair-density functional theory predict the correct atomic charges for dissociation an

Brønsted Acid-Promoted C–H Bond Cleavage via Electron Transfer from Toluene Derivatives to a Protonated Nonheme Iron(IV)-Oxo Complex with No Kinetic Isotope Effect | Journal of the American Chemical Society
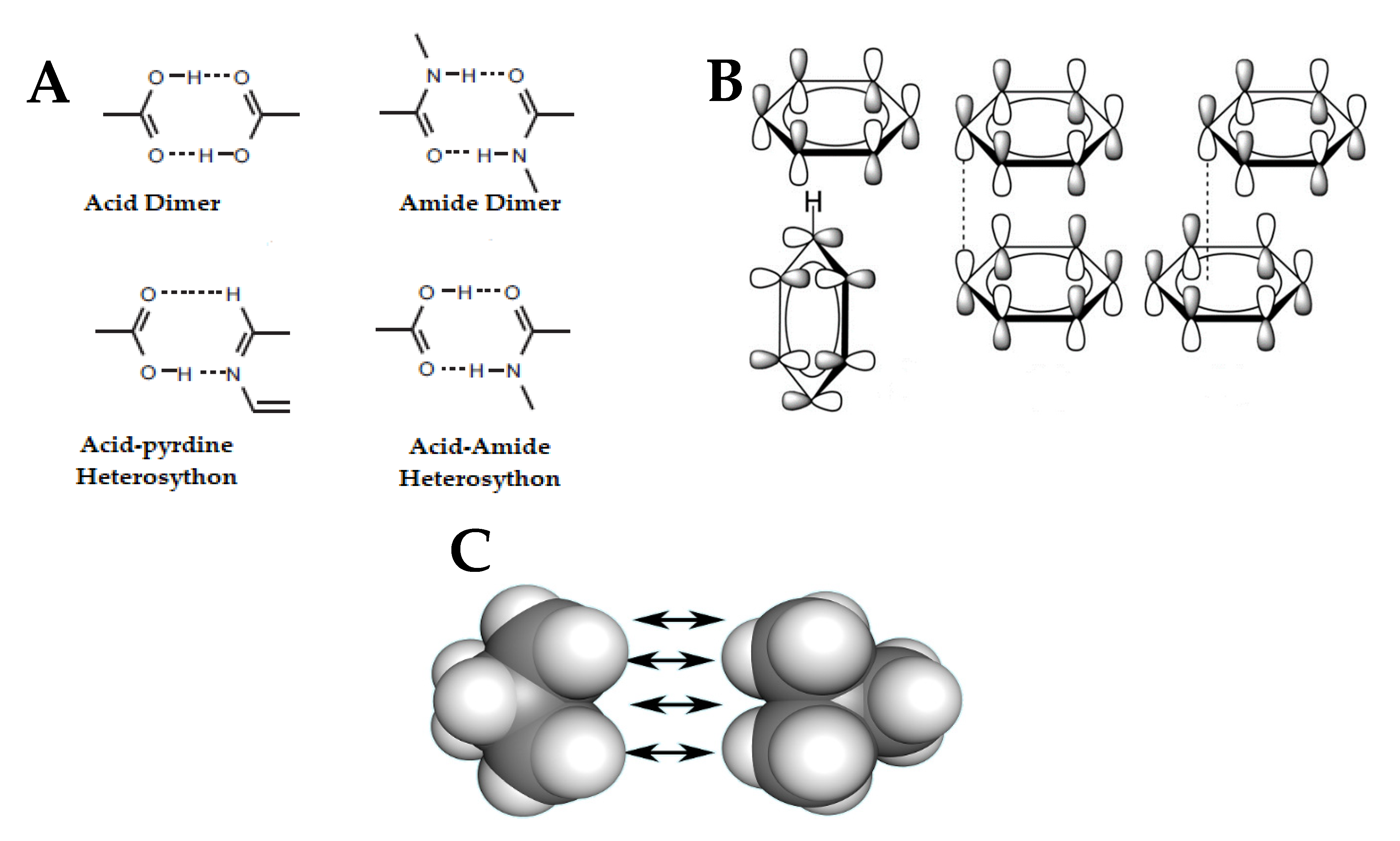
Crystals | Free Full-Text | Nano- and Crystal Engineering Approaches in the Development of Therapeutic Agents for Neoplastic Diseases | HTML
![PDF] Hydrogen Molecule Dissociation Curve with Functionals Based on the Strictly Correlated Regime. | Semantic Scholar PDF] Hydrogen Molecule Dissociation Curve with Functionals Based on the Strictly Correlated Regime. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/44d706e1233349d96782915ff4b7258f2aafc782/19-Figure3-1.png)
PDF] Hydrogen Molecule Dissociation Curve with Functionals Based on the Strictly Correlated Regime. | Semantic Scholar